FDA Nod for Wegovy Pill: A New Era in Obesity Treatment
The U.S. Food and Drug Administration has approved the first once-daily Wegovy pill for chronic weight management in adults with obesity or overweight, marking a milestone in obesity care with strong clinical results and broader access.
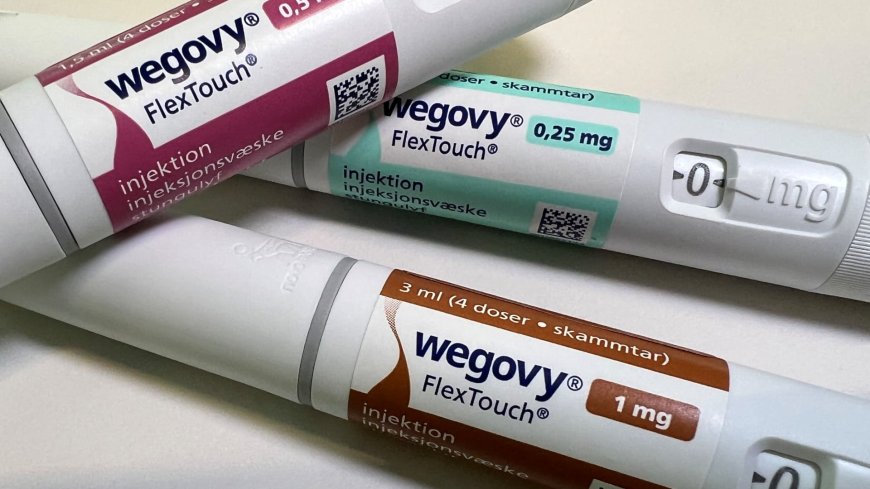
The U.S. Food and Drug Administration (FDA) has approved a once-daily pill version of Novo Nordisk’s Wegovy, making it the first oral GLP-1 weight-loss medicine approved in the United States for adults living with obesity or overweight with related medical conditions. weight loss
The active ingredient, semaglutide, is the same hormone-mimicking compound used in conventional Wegovy and Ozempic injections, which have been transformative in weight-loss and metabolic care.
Clinical Effectiveness and Benefits
Large clinical trials underpinning the approval showed strong weight-loss results:
-
Participants taking the 25 mg daily pill in the OASIS 4 trial lost on average about 16.6% of their body weight after 64 weeks when fully adherent.
-
Even without perfect adherence, weight loss averaged around 13.6%, demonstrating effectiveness comparable to the injectable version.
Nearly a third of patients in key trials achieved 20% or more weight loss, improving heart-health markers and physical activity outcomes.
Who Can Benefit
The approved Wegovy pill is indicated for:
-
Adults with obesity (BMI ≥ 30)
-
People overweight (BMI ≥ 27) with at least one weight-related medical condition, such as high blood pressure or type 2 diabetes
-
Adults with weight-related cardiovascular risk where both weight loss and heart-event risk reduction are therapeutic goals
The approval also includes the pill’s role in reducing the risk of major adverse cardiovascular events (like heart attack and stroke) in eligible adults—a critical benefit beyond weight reduction.
Daily Pill vs. Injectables: Accessibility & Convenience
Unlike weekly injections, the daily oral format offers:
-
Greater convenience for patients who are hesitant about injections;
-
No need for refrigeration;
-
Potential for wider adherence and patient preference.
The pill is expected to be launched as early as January 2026 in the U.S., with a starting price around $149 per month, significantly lower than many injectable alternatives.
Safety and Side-Effect Profile
The safety profile of the oral pill mirrors that of other semaglutide products, with common side effects including nausea, diarrhea, vomiting, and constipation reported in trials.
As with other GLP-1 medications, patients should discuss risks—such as gastrointestinal symptoms—with healthcare providers to determine suitability and dosage strategy.
Market Impact and Competitive Landscape
The approval marks a major competitive milestone for Novo Nordisk in the growing global obesity-drug market, positioning Wegovy’s oral version ahead of key rivals like Eli Lilly’s pending obesity pill under FDA review.
Analysts expect the oral weight-loss segment to expand rapidly, with oral medications projected to account for about 20% of the market by 2030, supported by convenience, lower cost, and broader patient uptake.
What This Means for Patients and Healthcare
Obesity affects a large proportion of adults in the U.S. and globally, and effective, easy-to-administer therapies have been scarce. The Wegovy pill’s approval not only expands treatment options but may also improve long-term health outcomes for millions seeking sustainable weight management solutions.
What's Your Reaction?
 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Angry
0
Angry
0
 Sad
0
Sad
0
 Wow
0
Wow
0